Gas
Contents
Key Stage 2
Meaning
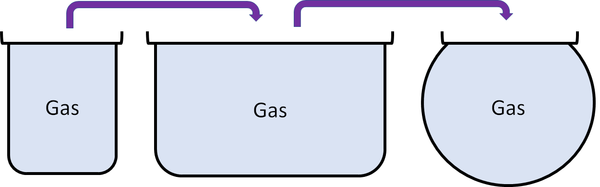
Gas is a state of matter that can change size and shape to fit any container.
About Gases
- Most gases are invisible but we can feel them.
- When the air moves we call it the wind.
|
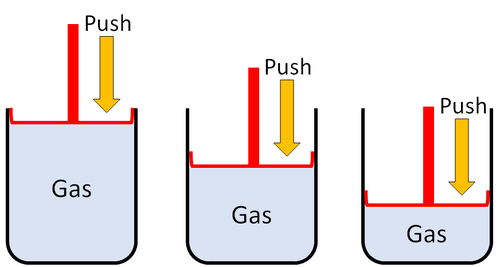
| Gases can be squashed into a smaller size. |
Examples of gas materials:
- Air (A mixture of gases, mostly nitrogen and oxygen)
- Steam
Key Stage 3
Meaning
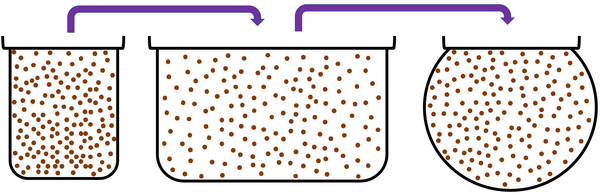
Gas is a State of Matter in which the particles are separated by large distances and can move freely.
About Gases
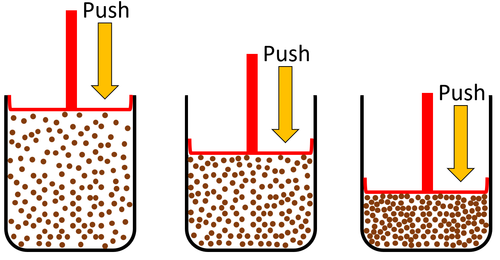
| Gas can be squashed into a smaller size because the particles are spread apart. |
Key Stage 4 Foundation
Meaning
Gas is a State of Matter in which the particles are separated by large distances and can move freely.
About Gases
- When a substance is in its gaseous state it is always less dense than in its liquid or solid state due to the particles in a gas being spread far apart from each other.
- A substance which is gaseous at room temperature has a smaller force of attraction between particles than a substance which is liquid or solid at room temperature.
| Particle Diagram | Particle Arrangement | Property |
| Particles are free to move in all directions. | Gases fit the size of their container. | |
| Gases fit the shape of their container. | ||
| Convection happens most easily in gases. | ||
| Particles are spread apart. | Gases can be compressed into a smaller volume. | |
| Sound passes through gases slower than liquids and solids. | ||
| Thermal Conduction is very poor in a gases. |
Key Stage 4 Higher
Molar Gas Volume
- At Standard Temperature and Pressure (20°C and 101,000Pa) the volume of one mole of gas is 24 dm3.
- The volume of gas required or produced in a chemical reaction can be calculated from the mass of the gas and its Relative Formula Mass.
Example Calculations
| Calculate the volume of 4g of Hydrogen gas. | Calculate the volume of 8g of Oxygen gas. | Calculate the volume of 20g of Methane gas. |
| 1.Find the Relative Formula Mass of the gas.
Chemical Formula of Hydrogen = H2 Relative Atomic Mass of Hydrogen = 1g Relative Formula Mass of H2 = 2g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Oxygen = O2 Relative Atomic Mass of Oxygen = 16g Relative Formula Mass of O2 = 32g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Methane = CH4 Relative Atomic Mass of Hydrogen = 1g
Relative Formula Mass of CH4 = 16g |
| 2. Calculate the number of moles of gas.
1 mole = 2g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{4}{2}\) No. Moles = 2mol |
2. Calculate the number of moles of gas.
1 mole = 32g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{8}{32}\) No. Moles = 0.25 mol |
2. Calculate the number of moles of gas.
1 mole = 16g No. Moles = \(\frac{Mass}{M_r}\) No. Moles = \(\frac{20}{16}\) No. Moles = 1.25 mol |
| 3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (2) Volume = 48dm3 |
3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (0.25) Volume = 6dm3 |
3. Find the volume in dm3.
Volume = 24 x (number of moles) Volume = 24 x (1.25) Volume = 30dm3 |
| Calculate the mass of 12dm3 of Hydrogen gas. | Calculate the mass of 36dm3 of Nitrogen gas. | Calculate the mass of 18dm3 of Ethane gas. |
| 1.Find the Relative Formula Mass of the gas.
Chemical Formula of Hydrogen = H2 Relative Atomic Mass of Hydrogen = 1g Relative Formula Mass of H2 = 2g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Nitrogen = N2 Relative Atomic Mass of Nitrogen = 14g Relative Formula Mass of N2 = 28g |
1.Find the Relative Formula Mass of the gas.
Chemical Formula of Ethane = C2H6 Relative Atomic Mass of Hydrogen = 1g Relative Atomic Mass of Carbon = 12g Relative Formula Mass of C2H6 = 30g |
| 2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 12 = 24 x (number of moles) Number of moles = 12/24 Number of moles = 0.5 |
2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 36 = 24 x (number of moles) Number of moles = 36/24 Number of moles = 1.5 |
2. Calculate the number of moles of gas.
Volume = 24 x (number of moles) 12 = 24 x (number of moles) Number of moles = 18/24 Number of moles = 0.75 |
| 3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 0.5 = \(\frac{Mass}{2}\) Mass = \(0.5\times2\) Mass = 1g Volume = 48dm3 |
3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 1.5 = \(\frac{Mass}{28}\) Mass = \(1.5\times28\) Mass = 42g |
3. Find the mass of gas from the number of moles.
No. Moles = \(\frac{Mass}{M_r}\) 0.75 = \(\frac{Mass}{30}\) Mass = \(0.75\times30\) Mass = 22.5g |
References
AQA
- Gas, pages 13, 34-5, 82-5, 96-101, GCSE Physics; Student Book, Collins, AQA
- Gas; pressure, pages 82-5, 96-9, 257, GCSE Physics; Student Book, Collins, AQA
- Gas; volume, page 98, GCSE Physics; Student Book, Collins, AQA
- Gases pages 36, 37, 46, 107, GCSE Chemistry; The Revision Guide, CGP, AQA
- Gases, page 107, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Gases, page 71, GCSE Physics, Hodder, AQA
- Gases, page 88, GCSE Chemistry; The Revision Guide, CGP, AQA
- Gases, page 97, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Gases, pages 100, 101, 124, GCSE Chemistry, CGP, AQA
- Gases, pages 164-5, 323, GCSE Combined Science Trilogy 1, Hodder, AQA
- Gases, pages 6, 36-37, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Gases, pages 78-79, 83, 86-89, GCSE Physics; Third Edition, Oxford University Press, AQA
- Gases, pages 98, 99, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Gases; analysis, pages 184-185, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Gases; atmosphere, pages 194-205, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Gases; density of, page 323, GCSE Combined Science Trilogy 1, Hodder, AQA
- Gases; gas pressure, page 193, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; gas pressure, page 41, GCSE Physics; The Revision Guide, CGP, AQA
- Gases; gas syringes, pages 140, 232, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; greenhouse effect, pages 198-202, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Gases; Greenhouse gases, pages 170-3, GCSE Combined Science Trilogy 2, Hodder, AQA
- Gases; identification of, page 207, GCSE Chemistry, Hodder, AQA
- Gases; Identification of, pages 160, GCSE Combined Science Trilogy 2, Hodder, AQA
- Gases; In the atmosphere, pages 167, GCSE Combined Science Trilogy 2, Hodder, AQA
- Gases; measuring volumes, pages 140, 232, 237, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; natural gas, page 178, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; natural gas, pages 18, 21, GCSE Physics; The Revision Guide, CGP, AQA
- Gases; natural gas, pages 45, 46 54, 56, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Gases; natural gas, pages 47, 48, 56, 58, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Gases; particle model of, pages 330-1, GCSE Combined Science Trilogy 1, Hodder, AQA
- Gases; particle model of, pages 78-80, 85-6, GCSE Physics, Hodder, AQA
- Gases; particle motion, page 104, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Gases; particle motion, page 193, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; particle motion, pages 114, 115, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Gases; particle motion, pages 38, 41, GCSE Physics; The Revision Guide, CGP, AQA
- Gases; pressure, page 104, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Gases; pressure, page 36, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Gases; pressure, pages 114, 115, 169, 171, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Gases; states of matter, pages 106, 107, 110-112, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Gases; states of matter, pages 121, 122, 193, 195, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; states of matter, pages 38-40, GCSE Physics; The Revision Guide, CGP, AQA
- Gases; states of matter, pages 96, 97, 100-102, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Gases; tests for, page 153, GCSE Combined Science; The Revision Guide, CGP, AQA
- Gases; Volume and mass measurement, pages 120, GCSE Combined Science Trilogy 2, Hodder, AQA
- Gases; volume, pages 78-79, 129, GCSE Chemistry; Third Edition, Oxford University Press, AQA
Edexcel
- Gases, molar volumes of, pages 118-119, GCSE Chemistry, Pearson, Edexcel
- Gases, page 300, GCSE Physics, CGP, Edexcel
- Gases, pages 34, 35, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Gases, pages 96, 98, GCSE Chemistry, CGP, Edexcel
- Gases, pages 97, 98, GCSE Combined Science; The Revision Guide, CGP, Edexcel
- Gases; doing work, page 312, GCSE Physics, CGP, Edexcel
- Gases; pressure, page 424, GCSE Combined Science, Pearson Edexcel
- Gases; pressure, pages 192, 194-195, GCSE Physics, Pearson Edexcel
- Gases; pressure, pages 310-312, 319, GCSE Physics, CGP, Edexcel
- Gases; temperature, page 192, GCSE Physics, Pearson Edexcel
- Gases; temperature, page 424, GCSE Combined Science, Pearson Edexcel
- Gases; volume, pages 194-195, GCSE Physics, Pearson Edexcel
OCR
- Gases, page 12, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR
- Gases, page 18, Gateway GCSE Chemistry, Oxford, OCR
- Gases, pages 14, 17, 18, Gateway GCSE Physics; The Revision Guide, CGP, OCR
- Gases, pages 82, 152, 155, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR
- Gases; atmosphere, pages 252-257, Gateway GCSE Chemistry, Oxford, OCR
- Gases; calculating amounts, pages 170-171, Gateway GCSE Chemistry, Oxford, OCR
- Gases; changing state, pages 76-77, Gateway GCSE Chemistry, Oxford, OCR
- Gases; collecting, pages 128, 222, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR
- Gases; collection, pages 265, 267, Gateway GCSE Chemistry, Oxford, OCR
- Gases; detection, pages 146-147, 265, 267, Gateway GCSE Chemistry, Oxford, OCR
- Gases; pressure in, pages 17, 18, Gateway GCSE Physics; The Revision Guide, CGP, OCR
- Gases; pressure, page 179, Gateway GCSE Chemistry, Oxford, OCR
- Gases; refined crude oil, page 239, Gateway GCSE Chemistry, Oxford, OCR
- Gases; volume, pages 170-171, 174, Gateway GCSE Chemistry, Oxford, OCR