Particle
Contents
Key Stage 3
Meaning
A particle is a small piece of matter.
About Particles
- Particles are often made of other, smaller, particles.
- Particles are usually shown as round in shape but may be made of smaller particles making their shape a combination of the shapes of other particles stuck together.
Examples
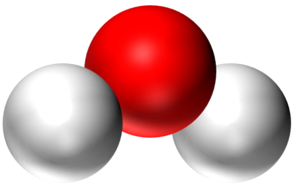
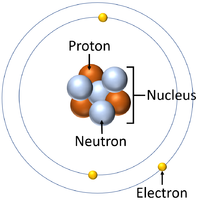
| A water molecule is a particle made of 1 Oxygen atom and two Hydrogen atoms. | An atom is a particle. | The atom is made of smaller particles called Protons, Neutrons and Electrons. |
Key Stage 4
Meaning
A particle is a small piece of matter.
About Particles
- Particles are often made of other, smaller, particles.
- Particles are usually shown as round in shape but may be made of smaller particles making their shape a combination of the shapes of other particles stuck together.
Examples
| A water molecule is a particle made of 1 Oxygen atom and two Hydrogen atoms. | An atom is a particle. | The atom is made of smaller particles called Protons, Neutrons and Electrons. |
References
AQA
- Particles, pages 48-111, GCSE Physics; Third Edition, Oxford University Press, AQA
- Particles; electric circuits, pages 50-63, GCSE Physics; Third Edition, Oxford University Press, AQA
- Particles; electricity in the home, pages 64-75, GCSE Physics; Third Edition, Oxford University Press, AQA
- Particles; kinetic theory of matter, page 79, 82-83, GCSE Physics; Third Edition, Oxford University Press, AQA
- Particles; molecules and matter, pages 76-91, GCSE Physics; Third Edition, Oxford University Press, AQA
- Particles; radioactivity, pages 92-111, GCSE Physics; Third Edition, Oxford University Press, AQA
Edexcel
Key Stage 5
Meaning
Particle is a general term used to describe a 'small' unit of matter.
About Particles
- The term particle may be used to describe objects as large as several billion atoms (as in a soot particle) or as small as a smallest possible unit of matter known as a fundamental particle.
- In physics the term particle is used:
- On larger scales to simplify calculations of the motions of objects by approximating them to be round with an even distribution of mass
- On large groups of objects at smaller scales for calculations of bulk properties by approximating them to be points with negligible volume.
- On atomic and subatomic scales to name units of matter which are often repeated in nature (eg. the proton is a repeating unit of matter found in all atoms).