Reactivity
Contents
Key Stage 4
Meaning
Reactivity is how vigorously a chemical will react.
About Reactivity
- Reactivity is determined by how easily an element can lose or gain electrons.
- Electrons are held in orbit around the nucleus because the electrons are negatively charged and are attracted to the nucleus which is positively charged.
- If an element loses electrons easily it is highly reactive.
- If an element gains electrons readily it is also highly reactive.
Three important factors affect reactivity of elements.
- The charge of the nucleus
- The shielding effect of inner electrons.
- Distance between the nucleus and the outer shell.
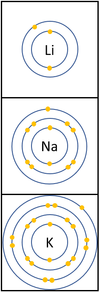
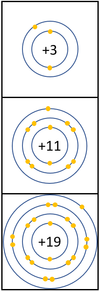
Reactivity in Groups 1, 2 and 3
| In a chemical reaction the electron in the outer shell is lost.
The reactivity increases as you go down the group because:
|
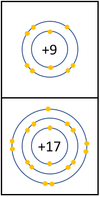
Reactivity in Group 7
| In a chemical reaction an extra electron is added to the outer shell.
The reactivity decreases as you go down the group because:
|
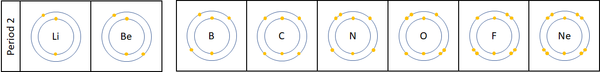
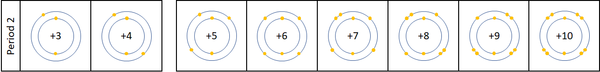
Reactivity along Period 2
| For the first 3 elements Lithium, Beryllium and Boron all lose electrons in chemical reactions.
The reactivity decreases as you go across the period because:
Nitrogen, Oxygen and Fluorine can all gain electrons to become negative ions in certain reactions. The reactivity increases as you go across the period because:
|
References
AQA
- Reactivity; alkali metals, pages 26-27, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Reactivity; metals, pages 26-27, 84-89, 220-221, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Reactivity; periodicity, pages 19, 30-31, GCSE Chemistry; Third Edition, Oxford University Press, AQA
Edexcel
OCR
- Reactivity; metals, pages 70-71, 125, 133, 141-143, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; noble gases, page 137, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; practical activities, pages 264-265, Gateway GCSE Chemistry, Oxford, OCR
- Reactivity; trends in Periodic Table, pages 133, 135, 137, 142-143, Gateway GCSE Chemistry, Oxford, OCR