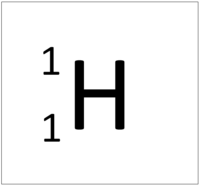
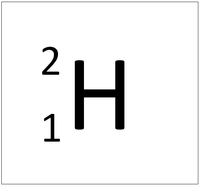
Isotope
Contents
Key Stage 4
Meaning
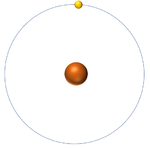
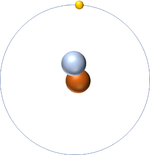
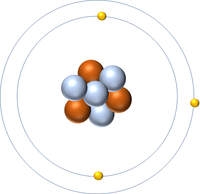
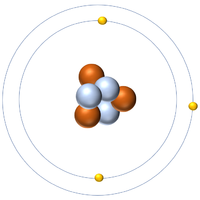
Isotopes are atoms of the same element (have the same number of protons) but different numbers of neutrons.
About Isotopes
- Each element has many isotopes but some are stable so they will exist for billions of years and others are unstable so they decay quickly into other isotopes or a different element.
- Different isotopes of the same element have the same atomic number but different atomic mass due to the different numbers of neutrons.
- Different isotopes of the same element have the same chemical properties but they have different physical properties. This means they cannot be separated by chemical processes but they can be separated by physical ones.
- Different isotopes of the same element may have different boiling points, different melting points, may diffuse at different rates and, as has already been stated, have a different mass. This means isotopes can be separated by processes such as Distillation, Chromatography and Centrifuge.
Examples
| Hydrogen-1 | Hydrogen-2 | Lithium-7 | Lithium-6 |
| Hydrogen always has 1 proton but isotope there are no neutrons. | Hydrogen always has 1 proton but in this isotope there is 1 neutron. This isotope of Hydrogen is known as Deuterium. | Lithium always has 3 protons but in this isotope there are 4 neutrons. | Lithium always has 3 protons but in this isotope there are 3 neutrons. |
Isotopic Abundance
- Different isotopes of the same element do not all appear in the same quantities. Some isotopes are more common than others.
- The Periodic Table orders the elements due to their chemical properties but since isotopes of the same element have the same chemical properties then different isotopes are not included.
- On the Periodic Table an average atomic mass is given based on the mass of each isotope and how common it is.
- To find the average atomic mass the mass of each isotope is considered along with the ratio of abundance.
Calculating the Average Relative Atomic Mass
| Chlorine has two common stable Isotopes; Chlorine-35 and Chlorine-37. Chlorine-35 is three times more common than Chlorine-37 so the ratio is 3:1. Find the average atomic mass of Chlorine. | Lithium has two common stable Isotopes; Lithium-7 and Lithium-6.
92.5% of all Lithium is the isotope Lithium-7 while Lithium-6 makes up only 7.5%. Find the average atomic mass of Lithium. |
| 1. State the known quantities.
Ratio: 3:1 3xChlorine-35 : 1xChlorine-37 |
1. State the known quantities.
Ratio: 92.5:7.5 92.5xLithium-7 : 7.5xLithium-6 |
| 2. Substitute the numbers into the equation and solve.
To find the average\[Average=\frac{35+35+35+37}{4}\] \(Average=\frac{35 \times 3 + 37 \times 1}{4}\) \(Average=35.5\) So the Relative Atomic Mass of Chlorine is quoted as 35.5 on the Periodic Table. |
2. Substitute the numbers into the equation and solve.
To find the average\[Average=\frac{7 \times 92.5 + 6 \times 7.5}{100}\] \(Average=6.925\) So the Relative Atomic Mass of Lithium is quoted as 6.9, 6.93 or 6.925 depending on the precision of that particular Periodic Table. |
References
AQA
- Isotope, pages 109-10, 117, GCSE Physics; Student Book, Collins, AQA
- Isotope; definition, page 110, GCSE Physics; Student Book, Collins, AQA
- Isotopes, page 112, GCSE Combined Science Trilogy; Physics, CGP, AQA
- Isotopes, page 124, GCSE Physics; The Complete 9-1 Course for AQA, CGP, AQA
- Isotopes, page 17, GCSE Chemistry; Third Edition, Oxford University Press, AQA
- Isotopes, page 44, GCSE Physics; The Revision Guide, CGP, AQA
- Isotopes, page 90, GCSE Physics, Hodder, AQA
- Isotopes, pages 119-20, 340, GCSE Combined Science Trilogy 1, Hodder, AQA
- Isotopes, pages 13, 21, GCSE Chemistry; The Revision Guide, CGP, AQA
- Isotopes, pages 25, 26, 51, GCSE Chemistry, CGP, AQA
- Isotopes, pages 27, 28, 51, GCSE Combined Science Trilogy; Chemistry, CGP, AQA
- Isotopes, pages 4-5, GCSE Chemistry, Hodder, AQA
- Isotopes, pages 96-97, 104-107, 109, GCSE Physics; Third Edition, Oxford University Press, AQA
- Isotopes, pages 97, 106, 198, GCSE Combined Science; The Revision Guide, CGP, AQA
Edexcel
- Isomers, page 173, GCSE Chemistry, Pearson, Edexcel
- Isotopes, page 155, GCSE Physics, CGP, Edexcel
- Isotopes, page 17, GCSE Chemistry; The Revision Guide, CGP, Edexcel
- Isotopes, page 93, GCSE Physics, Pearson Edexcel
- Isotopes, pages 166-167, GCSE Combined Science, Pearson Edexcel
- Isotopes, pages 22-23, GCSE Chemistry, Pearson, Edexcel
- Isotopes, pages 357, GCSE Combined Science, Pearson Edexcel
- Isotopes, pages 37, 38, GCSE Chemistry, CGP, Edexcel
- Isotopes, pages 51, 53, GCSE Physics; The Revision Guide, CGP, Edexcel
- Isotopes, pages 80, 174, 176, GCSE Combined Science; The Revision Guide, CGP, Edexcel
OCR
- Isotopes, pages 15, 31, Gateway GCSE Chemistry; The Revision Guide, CGP, OCR
- Isotopes, pages 170-171, 183, Gateway GCSE Physics, Oxford, OCR
- Isotopes, pages 28-29, Gateway GCSE Chemistry, Oxford, OCR
- Isotopes, pages 74, 77, 79, Gateway GCSE Physics; The Revision Guide, CGP, OCR
- Isotopes, pages 86, 195, Gateway GCSE Combined Science; The Revision Guide, CGP, OCR